Published at Rejuvenation Science News
Abstract
Extracellular vesicles (EVs) are nanometer-sized, lipid membrane–enclosed vesicles secreted by most, if not all, cells and contain lipids, proteins, and various nucleic acid species of the source cell. EVs act as important mediators of intercellular communication that influence both physiological and pathological conditions. Given their ability to transfer bioactive components and surmount biological barriers, EVs are increasingly being explored as potential therapeutic agents. EVs can potentiate tissue regeneration, participate in immune modulation, and function as potential alternatives to stem cell therapy, and bioengineered EVs can act as delivery vehicles for therapeutic agents. Here, we cover recent approaches and advances of EV-based therapies.
Extracellular vesicles (EVs) comprise a highly conserved and advanced system of intercellular communication, by which cells can exchange information in the form of lipids, proteins, or nucleic acid species. EVs were originally found to be involved in bone mineralization, as well as platelet function, and were called “platelet dust” (1). In the early 1980s, two separate publications described that exosomes, a subtype of EVs, can also help discard molecules that a cell does not need (2, 3). In these studies, reticulocytes expelled transferrin receptor in exosomes during their maturation to erythrocytes. Subsequent studies in the 1990s showed that EVs were highly regulatory in the immune system (4), and another decade later, it became evident that they were also able to shuttle proteins and RNA between cells (5–7). Over the past 5 years, research has started to shed light on the various mechanisms by which EVs can regulate biological functions, which span from tissue homeostasis and regulation of inflammation to the growth and metastasis of tumors. In view of their exceptionally broad biological functions and their ability to shuttle large molecules between cells, EVs offer a unique platform for the development of a new class of therapeutics.
EVs are present in all body fluids and are released by all types of cells in the human body. Classically, EVs have been divided into exosomes, smaller vesicles that are released from the interior of any cell via the multivesicular endosomal pathway, and microvesicles that are released from cells by budding of its surface membrane (8, 9). A third, less studied subgroup of EVs, known as apoptotic bodies, are formed by blebbing of dying cells and may contain diverse parts of the cell (10). In this Review, we focus on the first two classes of EVs. Until now, scientists based these classifications on EVs prepared by differential centrifugation, with “microvesicles” typically being isolated by a 10,000g to 20,000g centrifugation and the “exosomes” by a very high speed centrifugation at or above 100,000g (11). Preparations of microvesicles and exosomes are different in many ways, although there are overlaps in size and content (12). They contain distinct proteins and RNA cargo, which suggests that they mediate various biological functions through different molecular mechanisms. Current research indicates that further subdivisions of EVs may be needed to differentiate subtypes, for example, mitochondrial protein-enriched EVs (13) and different types of exosomes (12).
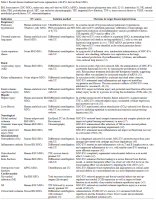
When developing an EV therapeutic, the first consideration is the cellular source. Thus, EVs from inflammatory cells naturally mediate different biological functions than EVs from mesenchymal stromal cells (MSCs). Multiple efforts are now ongoing in developing MSC-EVs as therapeutics, and multiple experimental studies report that EVs from MSCs mimic the immunoregulatory function and the regenerative capacity of MSCs (Table 1). Culture conditions, yield, and manufacturability are important aspects to consider that will be discussed in this Review but also are extensively discussed in another recent review (14). To overcome issues related to mammalian cell EVs, several research groups have also started to manufacture EVs from different types of fruit or vegetables, including ginger, grapes, and lemons (15–17), and it has been shown that these can be loaded with small molecular cargos, such as methotrexate, and mediate therapeutic effects in animal models (18).
Table 1 Recent disease treatment and tissue regeneration with EVs derived from MSCs.
BM, bone marrow; ESC-MSCs, embryonic stem cell–derived MSCs; hiPSCs, human induced pluripotent stem cells; IL-10, interleukin-10; NK, natural killer; PEG, polyethylene glycol; SEC, size exclusion chromatography; TFF, tangential flow filtration; TNF-α, tumor necrosis factor–α; VEGF, vascular endothelial growth factor; UC, ultracentrifugation.
Therapeutic EVs may also be modified by using molecular engineering techniques. Such engineered EVs may mediate biological functions in fundamentally different ways. EVs can be loaded exogenously by incorporating cargo on or in isolated EVs or endogenously, in which the cargo is introduced into or generated by the producer cell to exploit the cellular machinery for cargo sorting into EVs (Fig. 1). EVs could be loaded with therapeutic RNA molecules (19, 20) or proteins (21) to be delivered to the inside of recipient cells. Alternatively, therapeutic EVs could be engineered to express specific surface molecules, such as biologically active proteins that mediate a specific biological function or a molecule that can neutralize circulating bioactive molecules. Surface ligands can also be used to target EVs to specific recipient cell types, which can facilitate crossing of physiologic barriers, such as the blood-brain barrier (BBB), when targeting neurons (19). Other additions to the EV surface could enable fusion with the plasma membrane of the recipient cell or facilitate cytoplasmic release of cargo after endosomal uptake. Last, the route of administration of EVs influences their biodistribution (22), which needs to be considered when developing any therapeutic modality to be used in patients.

Over the last few decades, biological medications, such as monoclonal antibodies and cell therapies including chimeric antigen receptor (CAR) T cells, have achieved tremendous advances in managing disease. We will discuss here why we think EVs are likely to be the next breakthrough in medical treatment and why well-designed EV therapeutics may help to manage and cure disease.
Exogenously supplied MSCs derived from different tissues including bone marrow, adipose tissue, and umbilical cord confer therapeutic benefit in a variety of diseases and have achieved success particularly in tissue regeneration (23). The initial hypothesis that MSCs, through cellular differentiation, would replace damaged tissue was partially abandoned following observations that very few, if any, cells stably engraft in the host (24, 25). The therapeutic benefits were instead suggested to be imparted by the secretome of MSCs, a hypothesis that was strengthened by observations that MSC-CM (conditioned media) could achieve therapeutic efficacy similar to that realized by MSC administration in many paradigms (26, 27). This has led to the concept of using the MSC secretome (a mixture consisting of EVs and paracrine soluble factors that may be separated from or associated with the EVs) as an alternative to direct MSC therapy in regenerative medicine. The CM contains the MSC secretome, and the therapeutic efficacy of MSC-CM can be mainly attributed to the constituent EVs within (28). EVs derived from MSCs have been reported to have therapeutic potential in preclinical studies in diverse tissues and indications, including the treatment of diseases and regenerative medicine targeting the lungs (29–31), kidney (32–34), liver (35, 36), central nervous system (37–39), cartilage (40–42), bone (43, 44), and heart (28, 45) (Table 1). However, the therapeutic potential of MSC-EVs is still controversial because of the complexity of MSCs regarding tissue origin and cell culture conditions. In addition, the isolation and purity of EVs in relation to other factors in the MSC secretome, including the potential co-isolation of contaminating proteins and nucleic acids, may result in invalid conclusions of EV content and function. The underlying mechanisms attributed to the therapeutic action of MSC-EVs by the transfer of their cargo, as well as the triggering of signaling pathways via cell surface interactions, are diverse and include mitigating or eliciting immune responses, reducing inflammation, inhibiting apoptosis, minimizing oxidative stress, stimulating wound repair, and promoting angiogenesis, which together act to ultimately ameliorate the adverse effects of diseases, promote healing, and restore function (28–46).
The field has mainly focused on MSC-derived EVs. However, similar to MSCs, a number of different cell types with stem cell–like properties are associated with potential immunomodulatory effects that could be harnessed for therapeutic applications. EVs derived from other regenerative and immunomodulatory cell sources, such as amniotic epithelial cells, endothelial progenitor cells, embryonic stem cells, induced pluripotent stem cells, cardiosphere-derived cells, and dendritic cells (DCs), have been reported to mediate therapeutic effects in preclinical models of wound healing (47, 48), pulmonary fibrosis (49), vascular repair (50), myocardial infarction (51–53), and vaccination (54). The regenerative capacities and immunomodulatory effects of stem cells have been shown to be dependent on various factors, including donor-associated effects and tissue of origin (55, 56), and these effects may likely extend to their secreted EVs. In addition, culture conditions affect the composition and function of cells and their EVs. Cells exposed to stress-induced conditions, such as oxidative stress (57), acidic conditions (58), serum starvation (59), hypoxia (60), ultraviolet (UV) light (57), irradiation (61), or cell-stimulating substances (62), generate varying numbers of EVs with a different composition and function as compared to EVs isolated from cells under normal culture conditions. It is, however, questionable how representative the common cell flask culturing conditions are to physiologically relevant conditions. Three-dimensional (3D) cell culturing in bioreactors, on spheres, or in organoids, believed to mimic the physiological cell conditions better than 2D cultures, gives rise to EVs with altered properties compared to corresponding EVs derived from cells grown as monolayers on flat plastic dishes (63, 64).
Direct comparisons of the efficacy of parental cell therapy with EV administration are lacking, partly due to MSCs’ potential to provide a long-term source of EVs on site and the fact that EV injection may or may not be on site and/or recapitulate the number and length of action of EVs released by resident MSCs. MSC-EVs appear to be as effective as their parental MSCs in attenuating hyperoxic lung injuries or mitigating lung inflammation (30); however, others have shown minimal potency of MSC-EVs compared to MSC therapy for bone regeneration (65). Nevertheless, on the weight of the considerable evidence of their therapeutic utility amassed in preclinical studies, EVs are now being explored by various commercial entities for clinical translation. There are additional logistical advantages of using EVs, which can be considered as an off-the-shelf product, and EVs are also likely to have reduced potential side effects because they are less complex and better defined as compared to cell therapies.
The first report of native MSC-EV therapy in humans encompassed the treatment of one patient suffering from severe therapy-refractory acute graft-versus-host disease (GvHD) with EVs derived from four different bone marrow donors. The therapy was associated with improvement in clinical GvHD symptoms within the first week of MSC-EV therapy that remained stable 4 months after treatment (66). There have been few clinical studies conducted to date, which have evaluated native MSC-derived EVs. The first was a phase 1 clinical trial to evaluate human umbilical cord blood–derived MSC-EVs for the modulation of β cell mass in type 1 diabetes mellitus (ClinicalTrials.gov identifier NCT02138331); however, no information has been made available for this trial. The same team conducted a subsequent randomized, placebo-controlled, phase 2/3 clinical pilot study to investigate the safety and therapeutic efficacy of human cord blood–derived EVs in inhibiting the progression of grade III and IV chronic kidney disease (CKD). Outcomes showed that MSC-EV administration was safe, modulated the inflammatory immune reaction, and ameliorated overall kidney function in grade III-IV CKD patients (67). In addition, a phase 1 clinical trial to assess the safety and efficacy of MSCs and MSC-EVs for promoting healing of large and refractory macular holes (MHs) is currently ongoing (NCT03437759), and one further clinical trial is in the recruitment stage (NCT03437759). Although very early in terms of clinical use, together with evidence from preclinical studies, these clinical observations indicate that harnessing the innate ability of the MSC secretome by administration of one of the components, MSC-EVs, may hold promise for an acellular, off-the-shelf therapeutic strategy. An overview of clinical trials using EVs is shown in Table 2.
.....
Edited by Engadin, 16 May 2019 - 05:04 PM.