http://news.bbc.co.u...lth/7101834.stm
 L
onge
C
ity
Advocacy & Research for Unlimited Lifespans
L
onge
C
ity
Advocacy & Research for Unlimited Lifespans

Posted 20 November 2007 - 06:25 PM
Posted 20 November 2007 - 06:37 PM
So what are the differences between these reprogrammed cells and stem cells made by therapeutic cloning?
Posted 20 November 2007 - 08:04 PM
Posted 20 November 2007 - 08:07 PM
We are probably still 10 years from those break throughs and maybe more but it is definitely the direction things are going.
Posted 20 November 2007 - 08:26 PM
Do these cells get their "clock turned back" (i.e. telomere relenghtening) like ESCs derived from SCNT?
Posted 20 November 2007 - 11:59 PM
One rather relevant differences is that mice grown from cloned embryonic stem cells are just fine, while mice grown from these reprogrammed cells quickly die from cancers all over the place, presumably due to the overexpressed oncogenes (this is actual data, which for some reason these news releases don't consider worth mentioning).So what are the differences between these reprogrammed cells and stem cells made by therapeutic cloning?
Posted 21 November 2007 - 03:14 AM
Dolly demonstrated that there was something in the egg that could reverse the cell's "strong arrow of time," Thomson said.
Posted 21 November 2007 - 03:23 AM
One rather relevant differences is that mice grown from cloned embryonic stem cells are just fine, while mice grown from these reprogrammed cells quickly die from cancers all over the place, presumably due to the overexpressed oncogenes (this is actual data, which for some reason these news releases don't consider worth mentioning).
Posted 21 November 2007 - 04:25 AM
Posted 21 November 2007 - 04:34 AM
Introduction of the four 'Yamanaka factors' requires genetic manipulation using viral vectors that health agencies would be unlikely to approve for clinical use. And one of the factors, c-myc, is thought to be responsible for tumours in mice.
Thomson, who was the first to isolate and maintain human embryonic stem cells in culture, has gone part way towards solving these problems. He also used four factors, introduced by viral vectors, to reprogramme human foreskin cells. But only two of the four are the same, and he does not use c-myc. What is more, the discovery that a different recipe resulted in successful reprogramming suggests that scientists might have a greater degree of flexibility in finding clinically acceptable variations on Yamanaka's selection.
Furthermore, in a move likely to raise fresh controversy, Mitalipov this week began a collaboration with Alison Murdoch and Mary Herbert of Newcastle University, UK. Murdoch's group has a licence to work with human embryos. "I can't just keep modelling," Mitalipov says. "If we show medical progress, society will accept the technology."
Posted 21 November 2007 - 08:44 PM
Edited by G Snake, 21 November 2007 - 08:44 PM.
Posted 21 November 2007 - 10:12 PM
Edited by namingway, 21 November 2007 - 10:14 PM.
Posted 21 November 2007 - 11:19 PM
Posted 22 November 2007 - 04:50 AM
Edited by manofsan, 22 November 2007 - 04:52 AM.
Posted 22 November 2007 - 05:01 AM
Posted 22 November 2007 - 05:39 AM
... By finding ways to turn the knobs on genetic switches in the cell it was inevitable that scientists would figure out how to make cells change state into embryonic cells. They will next find more genetic knobs to turn in order to convert embryonic cells into precisely desired cell types and they will even find ways convert between various non-embryonic cell types while totally avoiding an intermediate state where the cells are like embryonic cells. Cells are just complex state machines. The next few decades of advance in biotechnology can be seen as a series of advances in techniques for causing desired and useful cell state transitions.
Posted 22 November 2007 - 10:05 AM
Posted 28 November 2007 - 01:13 AM
Posted 28 November 2007 - 01:34 AM
Posted 09 December 2007 - 12:45 AM
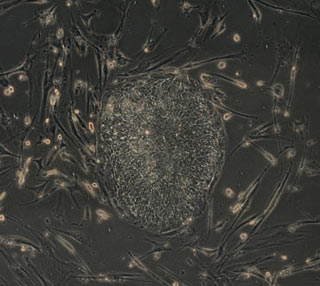
http://www.news.wisc...?clickcode=2923
UW-Madison scientists guide human skin cells to embryonic state
Nov. 20, 2007
by Terry Devitt
In a paper to be published Nov. 22 in the online edition of the journal Science, a team of University of Wisconsin-Madison researchers reports the genetic reprogramming of human skin cells to create cells indistinguishable from embryonic stem cells.
The finding is not only a critical scientific accomplishment, but potentially remakes the tumultuous political and ethical landscape of stem cell biology as human embryos may no longer be needed to obtain the blank slate stem cells capable of becoming any of the 220 types of cells in the human body. Perfected, the new technique would bring stem cells within easy reach of many more scientists as they could be easily made in labs of moderate sophistication, and without the ethical and legal constraints that now hamper their use by scientists.
The new study was conducted in the laboratory of UW-Madison biologist James Thomson, the scientist who first coaxed stem cells from human embryos in 1998. It was led by Junying Yu of the Genome Center of Wisconsin and the Wisconsin National Primate Research Center.
"The induced cells do all the things embryonic stem cells do," explains Thomson, a professor of anatomy in the University of Wisconsin School of Medicine and Public Health. "It's going to completely change the field."
In addition to exorcising the ethical and political dimensions of the stem cell debate, the advantage of using reprogrammed skin cells is that any cells developed for therapeutic purposes can be customized to the patient.
"They are probably more clinically relevant than embryonic stem cells," Thomson explains. "Immune rejection should not be a problem using these cells."
An important caveat, Thomson notes, is that more study of the newly-made cells is required to ensure that the "cells do not differ from embryonic stem cells in a clinically significant or unexpected way, so it is hardly time to discontinue embryonic stem cell research."
The successful isolation and culturing of human embryonic stem cells in 1998 sparked a huge amount of scientific and public interest, as stem cells are capable of becoming any of the cells or tissues that make up the human body.
The potential for transplant medicine was immediately recognized, as was their promise as a window to the earliest stages of human development, and for novel drug discovery schemes. The capacity to generate cells that could be used to treat diseases such as Parkinson's, diabetes and spinal cord injuries, among others, garnered much interest by patients and patient advocacy groups.
But embryonic stem cells also sparked significant controversy as embryos were destroyed in the process of obtaining them, and they became a potent national political issue beginning with the 2000 presidential campaign. Since 2001, a national policy has permitted only limited use of some embryonic stem cell lines by scientists receiving public funding.
In the new study, to induce the skin cells to what scientists call a pluripotent state, a condition that is essentially the same as that of embryonic stem cells, Yu, Thomson and their colleagues introduced a set of four genes into human fibroblasts, skin cells that are easy to obtain and grow in culture.
Finding a combination of genes capable of transforming differentiated skin cells to undifferentiated stem cells helps resolve a critical question posed by Dolly, the famous sheep cloned in 1996. Dolly was the result of the nucleus of an adult cell transferred to an oocyte, an unfertilized egg. An unknown combination of factors in the egg caused the adult cell nucleus to be reprogrammed and, when implanted in a surrogate mother, develop into a fully formed animal.
The new study by Yu and Thomson reveal some of those genetic factors. The ability to reprogram human cells through well defined factors would permit the generation of patient-specific stem cell lines without use of the cloning techniques employed by the creators of Dolly.
"These are embryonic stem cell-specific genes which we identified through a combinatorial screen," Thomson says. "Getting rid of the oocyte means that any lab with standard molecular biology can do reprogramming without difficulty to obtain oocytes."
Although Thomson is encouraged that the new cells will speed new cell-based therapies to treat disease, more work is required, he says, to refine the techniques through which the cells were generated to prevent the incorporation of the introduced genes into the genome of the cells. In addition, to ensure their safety for therapy, methods to remove the vectors, the viruses used to ferry the genes into the skin cells, need to be developed.
Using the new reprogramming techniques, the Wisconsin group has developed eight new stem cell lines. As of the writing of the new Science paper, which will appear in the Dec. 21, 2007 print edition of the journal Science, some of the new cell lines have been growing continuously in culture for as long as 22 weeks.
The new work was funded by grants from the Charlotte Geyer Foundation and the National Institutes of Health. In addition to Yu and Thomson, authors of the new study include Maxim A. Vodyanik, Kim Smuga-Otto, Jessica Antosiewicz-Bourget, Jennifer L. Frane and Igor I. Slukvin, all of UW-Madison; and Shulan Tian, Jeff Nie, Gudrun A. Jonsdottir, Victor Ruotti and Ron Stewart, all of the WiCell Research Institute.
More information: Learn more about stem cell and regenerative medicine research at UW-Madison by visiting the Stem Cell and Regenerative Medicine Center Web site.
http://www.stemcells.wisc.edu/
Posted 22 December 2008 - 08:42 PM
In a recent paper in Nature Structural and Molecular Biology, Professors Yehudit Bergman and Howard Cedar of the Hebrew University-Hadassah Medical School have deciphered the mechanism whereby embryonic cells stop being flexible and turn into more mature cells that can differentiate into specific tissues. Bergman is the Morley Goldblatt Professor of Cancer Research and Experimental Medicine and Cedar is the Harry and Helen L. Brenner Professor of Molecular Biology at the Medical School.
They found in their experiments, using embryos from laboratory mice and cells that grow in culture, that this entire process is actually controlled by a single gene, called G9a, which itself is capable of directing a whole program of changes that involves turning off a large set of genes so that they remain locked for the entire lifetime of the organism, thereby unable to activate any further cell flexibility.
Posted 22 December 2008 - 08:58 PM
Patient-derived induced stem cells retain disease traits
MADISON – When neurons started dying in Clive Svendsen's lab dishes, he couldn't have been more pleased.
The dying cells – the same type lost in patients with the devastating neurological disease spinal muscular atrophy – confirmed that the University of Wisconsin-Madison stem cell biologist had recreated the hallmarks of a genetic disorder in the lab, using stem cells derived from a patient. By allowing scientists the unparalleled opportunity to watch the course of a disease unfold in a lab dish, the work marks an enormous step forward in being able to study and develop new therapies for genetic diseases.
As reported this week in the journal Nature, Svendsen and colleagues at UW-Madison and the University of Missouri-Columbia created disease-specific stem cells by genetically reprogramming skin cells from a patient with spinal muscular atrophy, or SMA. In this inherited disease, the most common genetic cause of infant mortality, a mutation leads to the death of the nerves that control skeletal muscles, causing muscle weakness, paralysis, and ultimately death, usually by age two. (excerpt)
BBC on the same study
Posted 19 April 2009 - 10:50 PM
So what are the differences between these reprogrammed cells and stem cells made by therapeutic cloning?
In lay terms these cells are grown from your own cells so they bypass the need to insert your genome into a clone through STNC but they are still ASC (Adult Stem Cells) and their ability to be mutable into all the various types of cells that ESC (Embryonic Stem Cells) is still subject to a lot more study. However they have demonstrated a far greater plasticity than previous methods of manipulating ASC.
In a sense however they are still being reprogrammed, only using your own genotype as the base code so compatibility should be enhanced. The real trick will be developing methods of improved culturing that can not only harvest and cryo store your own stem cells but convert various tissues, skin, fat, and bone marrow for just a few, into significant quantities of matched stem cells, which can then be used to grow specific tissues in vitro while others portions of the harvest are stored.
We are probably still 10 years from those break throughs and maybe more but it is definitely the direction things are going.
Here is a another article on the same subject.
[url="http://www.cnn.com/2007/HEALTH/11/20/stem.cells.ap/index.html"http://www.cnn.com/2007/HEALTH/11/20/stem....s.ap/index.html"]http://www.cnn.com/2007/HEALTH/11/20/stem....s.ap/index.html[/url]
And another I posted previously in a different thread.
http://www.msnbc.msn.com/id/21886974/
Edited by crayfish, 19 April 2009 - 11:26 PM.
0 members, 1 guests, 0 anonymous users