Curious:
NR also elevated the mean concentration of nicotinamide (NaM), but this was not statistically significant (mean change = 106.5 pmol per mg protein; one-sided 95% CI (−10.03, ∞)). An increase in NaM would suggest an increase in the activity of NAD+-consuming enzymes, which catalyze the breakdown of NAD+ into NaM and ADP-Ribose.
In other studies (human and rodent), NR supplementation has consistently elevated NAM. Most of those studies have been acute studies, but it's still odd: you'd certainly expect it to be raised from first principles and existing data. It would certainly be disappointing and a challenge to the whole NAD+ precursor story if this were indeed a sign that NR does not increase the activity of NAD+-consuming enzymes such as SIRT1, SIRT3, SIRT6, and PARP1.
Consistent with the only other report of NR ingestion in humans, we were unable to detect NR concentrations in PBMCs during either treatment condition, despite using optimized recovery methods.
It's true that others have found this difficult: it's unfortunate that they didn't have contact with UWash or Imai (also coincidentally at UWash) to use better methods. Or maybe it's detectable in whole blood but not PBMC ...?
The magnitude by which NAD+ increased in response to NR supplementation was negatively associated with blood cellular NAD+ concentration during the placebo condition (R = −0.49, R2 = 0.25), suggesting a greater response in individuals with naturally low blood cellular NAD+ levels.
Very odd: UWash found "the rise in blood NAD+ (calculated as Cave,ss − baseline) following NR treatment did not correlate with the baseline blood level on Day 1 (R2 = 0.27, p = 0.2)" Moreover, the achieved level, as opposed to the relative amount of rise, was exactly the opposite to what they say here at Colorado:
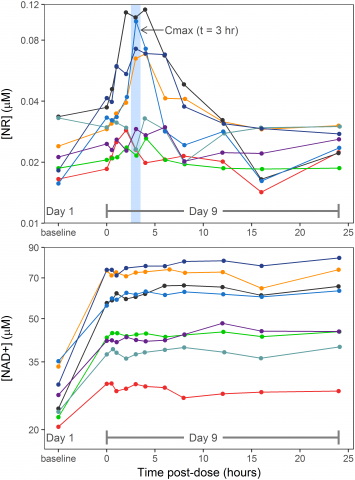
Figure 3, PLoS ONE 12(12): e0186459, PMID 29211728
Here we see reasonably clearly that those with the lowest baseline NAD+ also wound up with the lowest absolute post-supplementation NAD+ (and conversely for the highest) at the same dose and regimen at this trial.
we've had several clinical trials now, and none have reported changes in lipids or any aspects of glycemia. And 167 mg/dL is not a "very low level of cholesterol" — rather, as you also said in the next sentence, "It is on the lower normal range."
All my life i have seen only one person who has cholesterol at 160 range. The good results are in the 180 range and most are over 200.
You need to extend your social circles beyond the local Carl's Jr. 15% of the US population has TC ≤161 mg/dL; even in the 65-74 age group (the higher half of this trial), 10% of men and somewhat fewer women do.
Previous clinical trials have not tested cholesterol or A1C.
I did say "glycemia," not A1c per se. The Elysium, UWash, and now Colorado trials did test fasting glucose, and found no effect. Additionally, Colorado found no effect on fasting insulin, HOMA-IR (a calculated estimate of insulin sensitivity), HOMA-β (a calculated estimate of beta-cell function), and ostensibly found no effect on insulin sensitivity by IVGTT either, tho' it sure looks like NR actually worsened it: 4.53 ± 3.72 vs. 3.82 ± 1.88 (units not reported, but presumably 10-4 min-1 uU/mL) (Supplementary Table 7).
You need a population of people with abnormal levels to do it. Doing it on healthy people with normal levels will not show positive results.
Gotcha. All those healthy people taking NR should stop wasting their money, then.
MikeDC wrote: The unpublished clinical trial ChromaDex did probably showed some health benefit and they decided to publish the results in two papers. One on dose and safety and another on health benefit.
You have an amazing confidence about the results of studies that have not been conducted or published.































 This topic is locked
This topic is locked

























