.
A surveillance system in mammals constantly monitors cell activity to protect against aberrant proliferation in response to damage, injury and oncogenic stress. Here we isolate and culture connective tissue fibroblasts from highly regenerative mammals (Acomys and Oryctolagus) to determine how these cells interpret signals that normally induce cellular senescence in non-regenerating mammals (Mus and Rattus). While H2O2 exposure substantially decreases cell proliferation and increases p53, p21, p16, and p19 in cells from mice and rats, cells from spiny mice and rabbits are highly resistant to H2O2. Quantifying oxygen consumption and mitochondrial stability, we demonstrate that increased intracellular H2O2 is rapidly detoxified in regenerating species, but overwhelms antioxidant scavenging in cells from non-regenerative mammals. However, pretreatment with N-acetylcysteine (NAC) protects mouse and rat cells from ROS-induced cellular senescence. Collectively, our results show that intrinsic cellular differences in stress-sensing mechanisms partially explain interspecific variation in regenerative ability.
Introduction
Local cell proliferation is limited during tissue healing in most mammals, with new tissue forming a fibrous scar to repair the injury. During regeneration, resident cells respond to injury by actively proliferating to generate new tissue which subsequently undergoes morphogenesis and growth to replace the damaged or excised part1. While there is tremendous interest in understanding the source and developmental potential of the local progenitor cell population in regenerating systems (reviewed in ref. 2), it is equally important to understand how quiescent cells become activated and transduce injury signals to trigger cell cycle re-entry and controlled cell proliferation3. This last question is particularly relevant when trying to understand how closely related species can differ in their injury response; where active cell cycle progression and proliferation occur during regeneration, in contrast to cell cycle stasis and excessive collagen deposition during fibrotic repair4,5.
Although cell proliferation is tightly regulated during tissue homeostasis in adult vertebrates, a host of stimuli can lead to aberrant cell proliferation and neoplastic transformation of adult cells (reviewed in ref. 6). Cellular senescence has evolved as an important surveillance mechanism to identify damaged or mutated cells for subsequent removal or isolation. Originally identified to describe the finite proliferative capacity of human cells in culture7, a range of stimuli are now recognized to induce the stable cell-cycle arrest associated with cellular senescence (reviewed in refs. 8,9,10). Among these stimuli, tissue injury causes a local increase in reactive oxygen species (ROS) and can elicit a DNA-damage response (DDR) resulting in stress-induced or damage-induced senescence, respectively. During mammalian tissue repair, proper healing benefits from the secretory phenotype of senescent cells, although efficient clearing of senescent cells is also required for proper tissue remodeling11,12,13. Interestingly, results from several studies suggest that cells in regenerating vertebrates may be refractory to cellular senescence via alternate regulation of tumor suppressor pathways5,14,15,16. This raises the possibility that although injury-propagated cellular senescence might be beneficial for tissue repair in certain contexts, it may also prevent local cell proliferation and thus antagonize regenerative healing14,16,17,18.
Comparative models of regenerative success and failure provide an opportunity to investigate if intrinsic cellular differences can explain variation in regenerative ability19. Spiny mice (Acomys spp.) are capable of skin and musculoskeletal regeneration, whereas other related murid rodents heal identical injuries via fibrotic repair5,20,21,22,23. In response to a 4 mm ear punch assay, blastema formation and active cell proliferation distinguish the regenerative response in Acomys from scarring in Mus5. While local cells at the injury site were activated to re-enter the cell cycle in both species, significant cell cycle progression and proliferation only occurred during regeneration. Furthermore, injury in Mus induced nuclear localization of the tumor suppressor proteins p21 and p27 during fibrotic repair, whereas these tumor suppressors were not detected in Acomys blastemal cells5. In addition to spiny mice, certain lagomorphs can regenerate musculoskeletal tissue and are capable of producing at least 1 cm2 of new skin, cartilage, and connective tissue5,24,25,26. Although cell cycle progression and senescence have not been examined during regeneration in rabbits, alongside spiny mice, they provide an excellent opportunity to ascertain whether key cellular behaviors segregate across healing phenotypes.
In this study, we isolate and culture adult ear pinna fibroblasts from two highly regenerative (Acomys cahirinus and Oryctolagus cuniculus) and two non-regenerating (Mus musculus and Rattus norvegicus) mammals in order to assess proliferative ability and their propensity to experience cellular senescence using a comprehensive panel of consensus senescence markers. For clarity, we refer to these species by their genus throughout the remainder of the paper. First, we show that proliferative ability in culture does not correlate with healing phenotype across species. Next, we find that cells from non-regenerating species rapidly undergo cellular senescence in response to H2O2 and exhibit strong induction of the consensus markers p21, p53, p16, and p19 (ARF). In stark contrast, cells from regenerating species do not undergo stress-induced senescence in response to H2O2. While gamma irradiation induced DNA damage and senescence in all species, increased expression of p53 and p21 in Acomys and Oryctolagus was independent of p16 and p19. To mechanistically link ROS and stress-induced senescence we show that increased intracellular H2O2 is efficiently reduced via glutathione peroxidase (GPx) activity in regenerating species who do not exhibit mitochondrial distress. This contrasts to non-regenerating species which exhibit significant mitochondrial dysfunction in response to H2O2 exposure. Lastly, although exogenous ROS disrupts mitochondria and triggers cellular senescence in mouse and rat fibroblasts, we demonstrated that pretreatment with NAC protects these cells from ROS-induced cellular senescence.
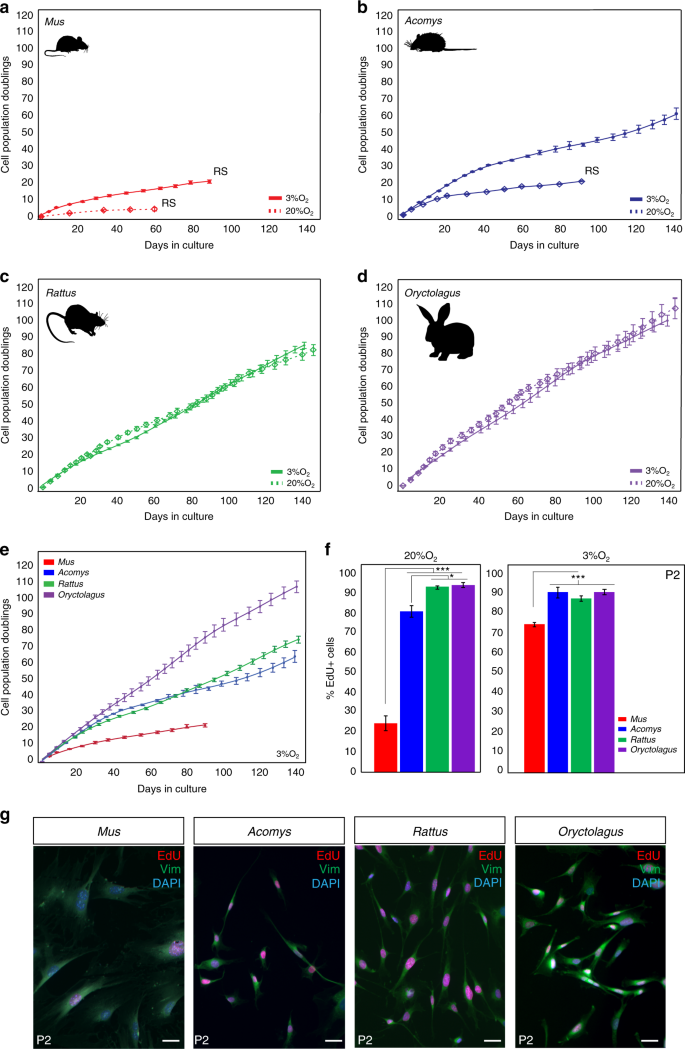
During vertebrate appendage regeneration, connective tissue fibroblasts are the dominant source for the local proliferative population that will replace the missing tissue27,28,29. To test our hypothesis that connective tissue fibroblasts from regenerating systems exhibit enhanced proliferative ability (compared to cells from non-regenerating animals), we isolated and cultured primary ear pinna fibroblasts from the highly regenerative Acomys and Oryctolagus and from two non-regenerating rodents Mus and Rattus under ambient or physiological oxygen levels (see Methods section). We and others have documented bonafide regeneration in spiny mice in refs. 5,16,20,21,30 and rabbits5,24,25, and rats have been shown to heal ear punches via fibrotic repair26. Under ambient oxygen, Mus fibroblasts entered stasis after ~43 days (mean population doublings (PDs) = 4.4) and Acomys fibroblasts appeared to senesce at ~90 days (PDs = 20.1) (Fig. 1a, b). We hypothesized that Rattus fibroblasts would behave similar to Mus, and Oryctolagus fibroblasts would exhibit enhanced proliferative capacity similar to Acomys. In fact, rabbit fibroblasts exhibited even higher proliferative capacity (107 PDs during 143 days in culture) compared to Mus and Acomys, while rat fibroblasts showed no signs of stasis even after 147 days in culture (PDs = 84.3) (Fig. 1c, d).
Fig. 1
Fibroblasts from Acomys, Rattus, and Oryctolagus exhibit enhanced proliferative ability. a–d PDs for Mus, Acomys, Rattus,and Oryctolagus fibroblasts cultured under ambient (20%) and physiological (3%) O2. a, b 3% O2 enhances proliferative capacity of Mus and Acomys fibroblasts: Mus (n = 5) cell lines at 3% O2: PDs = 20.8 ± 0.93, vs. 20% O2: PDs = 4.4 ± 0.97 and Acomys (n = 5) cell lines at 3% O2: PDs = 60 ± 3.4 vs. 20% O2: PDs = 20.1 ± 0.34. c–d O2 concentration did not affect mean proliferative ability of Rattus (n = 4) or Oryctolagus (n = 4) fibroblasts. e Cross-species comparison of proliferative ability at 3% O2 shows that Mus cells still senesce while fibroblasts from Acomys, Rattus, and Oryctolagus proliferate for at least 140 days. f At 20% O2, the proliferative population (EdU+) of P2 Mus cells (~25%) was significantly lower compared to Acomys (82%), Rattus (95%) and Oryctolagus (95%) (ANOVA, F = 143.3982, P < 0.0001) (n = 4/species). Percent EdU + cells in Acomys were slightly lower compared to Rattus (Tukey-HSD, t = −4.38, P = 0.0043) and Oryctolagus (Tukey-HSD, t = −4.87, P = 0.0019) (n = 4/species). At 3% O2, mean percent EdU + cells in P2 cultures are significantly lower in Mus (75%) compared to Acomys (88%), Rattus (91%) and Oryctolagus (91%) (ANOVA, F = 17.3085, Mus vs. Acomys, P = 0.0003, Mus vs. Rattus, P = 0.0023 and Mus vs. Oryctolagus, P = 0.0002) (n = 4/species). g P2 cells co-labeled with EdU and the general fibroblast marker Vimentin demonstrate that > 95% of cell cultures from all four species are fibroblasts (n = 4/species). Scale bars = 50 µm. Graphics for Acomys, Mus, and Rattus were made by corresponding author and the Oryctolagus image is available free for comercial use. ***P < 0.0001, **P < 0.001 and *P < 0.05. Error bars = S.E.M. Source data are provided as a Source Data file
Although fibroblasts from some mammals are insensitive to oxygen levels31, cells from other species (including humans) exhibit impaired proliferative capacity at ambient O2. To better approximate in vivo oxygen concentrations, we assayed cells under physiological O2 levels and asked whether reduced oxygen affected proliferative capacity in adult fibroblasts from Acomys, Mus, Rattus, and Oryctolagus (Fig. 1a–e). We found that physiological O2 significantly increased the proliferative ability of Mus and Acomys cells but had no effect on Rattus and Oryctolagus fibroblasts (Fig. 1a–e). Although Mus cells divided more under reduced oxygen (20.8 ± 0.93 PDs vs. 4.4 ± 0.97 PDs), they still experienced stasis rather quickly (~3 months in culture). Under 3% O2, Acomys fibroblasts divided for ∼60 PDs before exhibiting signs of reduced growth and grew at a similar rate to Rattus cells (Fig. 1b, e). In contrast to Mus and Acomys, oxygen concentration did not affect Rattus and Oryctolagus fibroblasts which exhibited almost identical growth rates after 5 months in culture at 20 and 3% O2 (Oryctolagus = 100 PDs, Rattus = 85 PDs) (Fig. 1c, d).
To support these observations, we estimated the proliferative population in culture by labeling passage 2 (P2) cells with EdU for 24 h under ambient and physiological oxygen (Fig. 1f). Under 20% O2, we found a significantly smaller proliferative population in Mus compared to Acomys, Rattus, and Oryctolagus (ANOVA, F = 143.39, P < 0.0001) (Fig. 1f and Supplementary Table 1). In addition, the total proliferative population of Acomys fibroblasts (82%) was slightly smaller compared to Rattus (95%) and Oryctolagus (95%) (Tukey-HSD, Acomys vs. Rattus t = −4.38, P = 0.0043; Acomys vs. Oryctolagus t = −4.87, P = 0.0019) (Fig. 1f). Similarly, at 3% O2, the percent of EdU + cells was not significantly different between Acomys (~88%), Rattus (~91%), and Oryctolagus (~91%), whereas the proliferative rate of Mus fibroblasts (~75%) was significantly lower compared to all three species (Fig. 1f and Supplementary Table 2). Alongside EdU, we used vimentin as a broad marker of fibroblast identity and found that our primary cell cultures contained >95% fibroblasts across all four species (Fig. 1g). Collectively, these data show that the intrinsic proliferative ability of ear pinna fibroblasts does not correlate with healing phenotype. In addition, reducing oxygen levels increases the proliferative capacity of Acomys and Mus cells, but has no effect on the population growth rate of Rattus and Oryctolagus cells.
.../...
F O R T H E R E S T O F T H E S T U D Y, P L E A S E V I S I T T H E S O U R C E .
.