.
The discovery that larger quantities of blood-borne proteins enter the brains of young, healthy mice than enter those of aged animals will alter our understanding of the blood–brain barrier, and how it changes with age.
The blood vessels in the brain have properties that limit their permeability to blood-borne ions, molecules and cells1. This blood–brain barrier (BBB) is crucial for proper neuronal function and for protecting the brain from harm, but it is also a major impediment to drug delivery1. It has been proposed that the BBB becomes more permeable with age, but, writing in Nature, Yang et al.2 find something quite different. They show that the BBB allows blood-borne proteins to enter the healthy brain at a much higher rate than previously thought, and that the overall amount of plasma proteins entering the brain actually decreases with age. The authors’ work could help researchers to understand how the brain responds to systemic protein signals, and the role of the BBB in age-related cognitive decline. It might also lead to improved approaches for delivering drugs into the brain.
The BBB is sometimes thought of as a static, impenetrable barrier. In reality, it has many dynamic properties — physical, transport, immune and more — that together tightly regulate the movement of molecules between the blood and the brain, thus controlling the brain’s molecular environment. A key question is s what exactly gets across the BBB?
Yang et al. addressed this by examining how the proteins found in blood plasma enter the brain. Whereas previous studies3–5 have traced the movement of injected, exogenous proteins (those not native to the organism), Yang and colleagues labelled endogenous mouse plasma proteins and injected them back into mice. In this way, they could track the movement of proteins that normally interact with the mouse BBB. They found that, in healthy young adult mice, a much higher quantity of plasma proteins than was previously thought enters the brain, and therefore has the potential to interact with the neuronal circuitry. This finding suggests that a wide variety of neural functions, including mood and behaviour, could be modulated by systemic protein signals.
The authors went on to show that the amount of plasma protein that permeates the brain is lower in old than in young mice. This was surprising because multiple studies that used exogenous tracers have shown that BBB permeability increases with age, and have highlighted this increase as a contributing factor to age-related cognitive decline6,7.
Yang et al. reconciled these seemingly disparate results by demonstrating age-related changes in the mechanism by which proteins are transported across the endothelial cells that line blood vessels of the BBB (Fig. 1). In young adults, the predominant method of transport involves the binding of specific proteins to plasma-membrane receptors on endothelial cells. These receptors become incorporated into vesicles and are transported across the cells — a process called receptor-mediated transcytosis. In aged mice, receptor-mediated transcytosis is significantly reduced and non-receptor-mediated (nonspecific) transcytosis increases, leading to nonspecific entry of a larger variety of plasma proteins into the brain. Previous studies that used exogenous molecules probably measured only nonspecific transcytosis, thus missing the vast majority of plasma-protein permeation into the young brain. The finding that the specificity of protein entry diminishes with age could indicate that ageing alters the brain’s ability to receive specific plasma-protein signals.
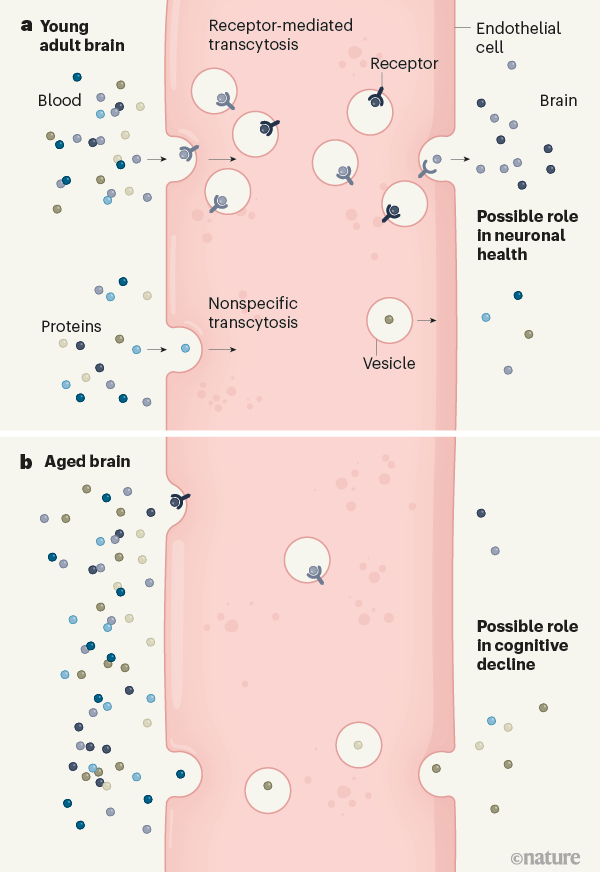
Figure 1 | Age-related changes in protein entry to the brain. Endothelial cells that line blood vessels of the brain restrict the entry of blood-borne proteins. However, some proteins cross these cells in vesicles. Yang et al.2 report that the methods by which proteins cross endothelial cells change with age. a, In young mice, the predominant mode of brain entry is receptor-mediated transcytosis, in which specific proteins that bind to receptors cross from the blood into the brain in vesicles. Less prominent is nonspecific transcytosis, in which proteins are incorporated into vesicles at random. b, In aged mice, the overall amount of protein that enters the brain is reduced. Nonspecific transcytosis increases and receptor-mediated transcytosis declines. It is possible that this switch is associated with age-related cognitive decline.
To understand the mechanism by which the BBB transports proteins, Yang et al. developed a method to correlate each endothelial cell’s level of plasma-protein uptake with its gene-expression profile, and analysed how this relationship changed along the vascular tree. This analysis revealed that there was a gradient in uptake of plasma proteins along the tree — from minimal on the arterial side (where blood arrives from the heart, and blood pressure is highest) to maximal on the venous side (where blood returns to the heart, and blood pressure is at its lowest). Thus, protein transport increases as the pressure in vessels decreases.
The authors also identified genes whose expression in endothelial cells correlates both positively and negatively with plasma-protein uptake. This list of genes might be useful for identifying transmembrane receptors involved in receptor-mediated transcytosis. Such receptors could be targets for ‘Trojan horse’ drug delivery8, in which proteins are engineered to bind to specific transmembrane receptors that can cross the BBB, such as the transferrin receptor. Because receptor-mediated transcytosis decreases with age, Yang and colleagues’ data indicate that the effectiveness of existing Trojan-horse methods (such as those based on the transferrin receptor) will also decrease with age. But the authors found that expression of the gene Alpl increases in brain endothelial cells in aged mice, and pharmacological inhibition of ALPL, the protein encoded by Alpl, increased receptor-mediated transcytosis of the transferrin receptor. This might therefore be a way to enhance Trojan-horse drug delivery, particularly in older people.
Yang and colleagues’ results provide remarkable insight into protein permeation into the brain. However, some limitations must be taken into account. For instance, the authors’ bulk protein-tracing experiments quantify protein permeation into the brain as a whole, but their imaging studies clearly reveal that levels of labelled proteins are higher in some brain regions than in others, including regions near the ventricles — the cavities that contain cerebrospinal fluid — and in the spaces immediately around some blood vessels. In addition to the BBB, plasma components can access the brain through barriers between blood and cerebrospinal fluid that are found in the choroid plexus of the ventricles and in the meninges that cover the brain. The relative contributions of each barrier to plasma-protein access to the brain remain unclear. It is also not known whether proteins access the entire brain or are restricted to certain compartments. Therefore, it is not clear to what extent plasma proteins interact with different neural circuits.













































