In Aging Cell, researchers have described how the enzyme Pck1, a core part of metabolic activity, is required for staving off senescence in fat (adipose) cells.
The aging of fat
With the decrease in metabolic activity that frequently occurs with aging, human beings often accumulate fat. This fat, itself, also ages; this paper describes it as “one of the most vulnerable tissues”, noting its association with physical problems [1] and its link to metabolic disorders, including insulin resistance [2]. Previous research has found that, in mice, clearing out senescent adipose cells mitigates some of these problems [3].
These researchers have done previous work demonstrating that phosphoenolpyruvate carboxykinase 1 (Pck1) deficiency shortens the lifespan of yeast [4]. As this enzyme is required for proper metabolic function in adipose tissue, the researchers sought to determine its relationship to senescence and aging in this context.

Read More
Necessary for mouse health
Unsurprisingly, as in many other tissues, 24-month-old mice were found to have more senescent fat cells (adipocytes) than 4-month-old mice. While overall Pck1 expression was largely restricted to mature adipocytes, the senescent cells expressed considerably less of it. Feeding mice a high-fat diet also decreased Pck1 expression.
The researchers then investigated the role of this enzyme by creating a strain of mice that does not express Pck1 in adipocytes. Compared to wild-type controls, these mice exhibited substantial increases in adipocyte senescence and metabolic issues, such as insulin resistance, at 12 and 24 months of age, with young mice being less affected; this was in spite of the mice not having any significant differences in body weight. A high-fat diet had even more negative effects on these modified mice than it had on wild-type mice.
The adipocytes of these modified mice were found to secrete substantially more SASP factors than those of unmodified mice, particularly at middle age. There was an increase of fibrosis as well along with more signs of immune cell infiltration, demonstrating increased inflammatory effects. Therefore, the researchers surmised that a lack of Pck1 in these cells exacerbates inflammaging, the age-related chronic inflammation that occurs even in the absence of pathogens.
Mitochondrial dysfunction and metabolic effects
Pck1 depletion led to a substantial increase in mitochondrial dysfunction. The affected cells had smaller and more misshapen mitochondria than unaffected cells. The mitochondria were also depolarized, showing a lack of functional ability, and they were afflicted by a rise in reactive oxygen species (ROS). A gene expression analysis found significant downregulations in key proteins needed for proper mitochondrial respiration.
This work identified four metabolites that accumulate in Pck1-depleted adipocytes: fumarate, succinate, glutamate, and DL-glutamate. All four are part of the TCA cycle, which is fundamental to cellular metabolism. Adding additional fumarate to these cells increased their expression of the senescence markers p21 and p16 even further. Further work targeting fumarate found that it was the key metabolite in these problems: removing it mitigated ROS and reduced the expression of the inflammatory cGAS/STING pathway, which had driven these cells senescent.
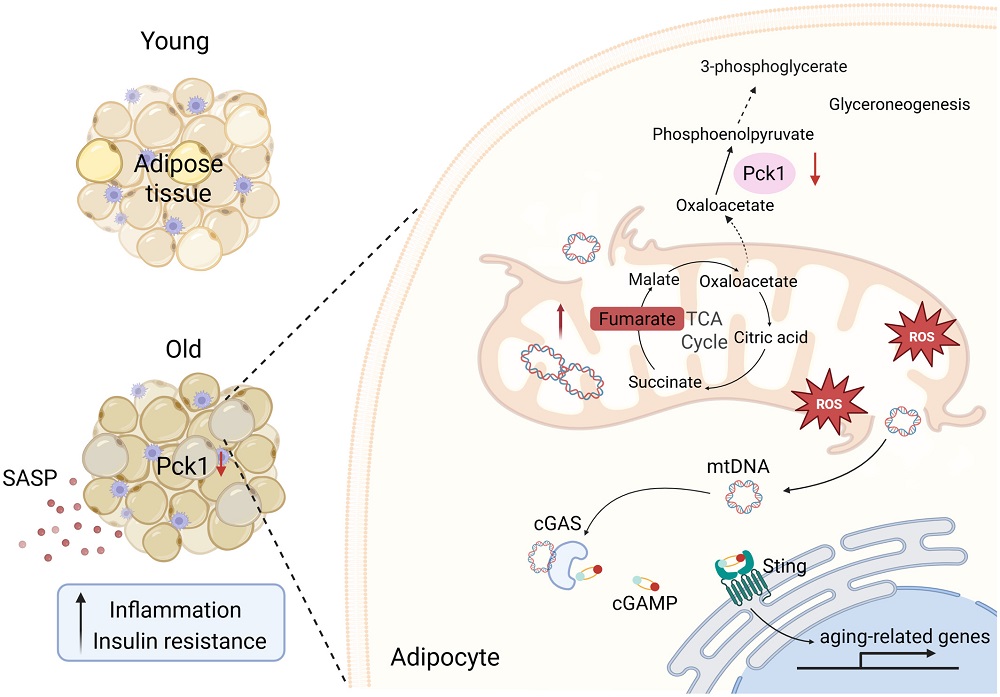
The researchers note that they do not know why Pck1 decreases in adipocytes with age, stating that this is a topic that they plan to investigate in further research. Similarly, they have not confirmed whether or not Pck1 is specific to adipocytes or if it applies to other tissues as well; they intend to use multi-organ models in order to discover this. However, they note that Pck1 is a “novel therapeutic target” and believe that targeting it may lead to effective treatments in the future.
Literature
[1] Ou, M. Y., Zhang, H., Tan, P. C., Zhou, S. B., & Li, Q. F. (2022). Adipose tissue aging: mechanisms and therapeutic implications. Cell death & disease, 13(4), 300.
[2] Reyes-Farias, M., Fos-Domenech, J., Serra, D., Herrero, L., & Sanchez-Infantes, D. (2021). White adipose tissue dysfunction in obesity and aging. Biochemical pharmacology, 192, 114723.
[3] de Oliveira Silva, T., Lunardon, G., Lino, C. A., de Almeida Silva, A., Zhang, S., Irigoyen, M. C. C., … & Diniz, G. P. (2025). Senescent cell depletion alleviates obesity-related metabolic and cardiac disorders. Molecular Metabolism, 91, 102065.
[4] Yuan, Y., Lin, J. Y., Cui, H. J., Zhao, W., Zheng, H. L., Jiang, Z. W., … & Liu, X. G. (2020). PCK1 deficiency shortens the replicative lifespan of Saccharomyces cerevisiae through upregulation of PFK1. BioMed Research International, 2020(1), 3858465.
View the article at lifespan.io








































