It seems to me that there is a simple trick to largely progress towards life extension:
TODAY
- Do you know how long standard lab mice live? ... about 2 years and a half on average; most die between 2 and 3 years old (similar with rats)
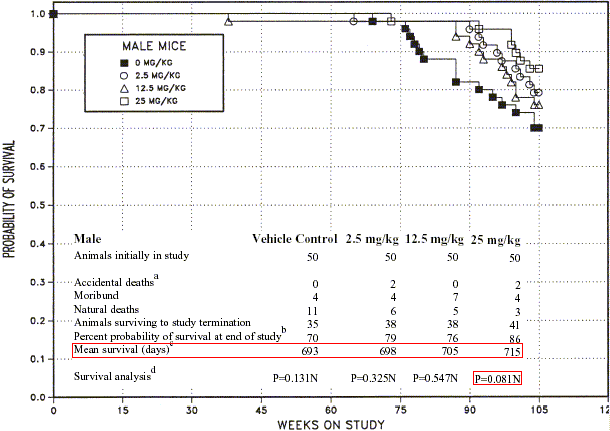
- Do you know how long toxicity/carcinogesis tests last in rodents? ... 2 years; probably because old age did not interest people when toxicity tests where initially designed
- Do you see the trouble? ... Because of these 6-12 months of test lacking,
for most drugs and chemicals that are around us* we have no idea of their impact on lifespan!!!
- Could it be different? ... Yes, technically it seems easy: simply don't stop toxicity/carcinogenesis tests so early. Everything is already in place, simply don't stop the experiment (at least when no severe drawbacks are found after 2 years; that's such a shame!)
- How to make it change? ... I'm not sure yet. Perhaps first contacting the NTP. Perhaps C.E.L. might want to participate. I thought it is such a specific subject with such a potential impact for life extension that it deserves its own thread. I'm still surprised that it might be so simple. I'm awaiting your reactions

Edited by AgeVivo, 01 July 2009 - 09:52 PM.