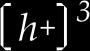Does oral SOD act more as an extracellular or intracellular agent ? I'm hoping it would substitute the function of extracellular SOD (SOD3)
Research documents provided by Glisodin seem to show SOD1. This is from Glisodin documents:
At the end of 21 days of supplementation, patients receiving GliSODin were found to have normalized circulating SOD1 activity and total antioxidant status, while no such effects were observed in the placebo or non-protected SOD groups. The researchers concluded that GliSODin could improve antioxidant defenses.
So I think that the idea is to make it function in the body like SOD3. But then there are some differences between the three types of SOD:
Super-SOD: superoxide dismutase chimera fights off inflammation
Andrew Gow and Harry Ischiropoulos
Paragraphs 2-3
The three SOD isomers, cytosolic Cu,Zn SOD (SOD1), mitochondrial MnSOD (SOD2), and extracellular Cu,Zn SOD (SOD3), have been shown to have some therapeutic utility in protecting organ systems from oxidative stress, particularly in animal model systems of disease. However, the success of these therapies has been limited due to a variety of reasons such as the short half-life of the protein in circulation, inability to associate with the cellular surface, and slow rates of equilibration between the vascular and interstitial spaces. Primarily due to the small molecular radius of SOD1 injected into circulation, it is rapidly (half-life of 10 min) cleared by the kidneys. Furthermore, its negative charge does not allow SOD1 to interact with cell surfaces and reduces its ability to enter the interstitium. Moreover, the therapeutic efficacy of SOD1 exhibits a bell-shaped curve after systemic administration, which, although not well understood, further limits the concentration of this protein that can be administered pharmacologically. These limitations are partially alleviated by the use of SOD2, which is the least negatively charged SOD, and in the tetrameric form has a molecular radius of 40 Å, which retards its clearance by the kidneys (plasma half-life of 4 h). Despite its larger size, SOD2 equilibrates nearly four times faster that SOD1 within interstitial spaces.
SOD3 is normally tagged to the cellular surface via its hydrophilic positively charged “tail”, which gives the protein its heparin-binding ability. Previously, it has been shown that cleavage of this tail results in the release of SOD3 from the cellular surface and that this loss may contribute to the sensitivity of the endothelium to oxidative insults. Furthermore, a major contributor of reactive intermediates near or at the endothelial plasma membrane is the NADPH oxidase. It is now recognized that a family of membrane-associated proteins (NOX) are responsible for generating superoxide and hydrogen peroxide in vascular endothelium and smooth muscle cells potentially for defense purposes and for cell signaling. The NOX enzymes appear to be composed of the typical low-potential membrane gp91phox flavoprotein that reduces oxygen to superoxide, as well as of cytosolic proteins, which in response to stimuli assemble into a functional oxidase. The generation of superoxide in the vascular compartment not only from activated inflammatory cells but also from vascular cells contributes to adverse effects of tissue injury during inflammation and other vascular disorders.
Paper link:
http://ajplung.physi...284/6/L915.full
Edited by Alec, 11 October 2011 - 07:18 AM.