.
F U L L T E X T S O U R C E : nature
Abstract
Bitter taste receptors play crucial roles in detecting bitter compounds not only in the oral cavity, but also in extraoral tissues where they are involved in a variety of non‒tasting physiological processes. On the other hand, disorders or modifications in the sensitivity or expression of these extraoral receptors can affect physiological functions. Here we evaluated the role of the bitter receptor TAS2R38 in attainment of longevity, since it has been widely associated with individual differences in taste perception, food preferences, diet, nutrition, immune responses and pathophysiological mechanisms. Differences in genotype distribution and haplotype frequency at the TAS2R38 gene between a cohort of centenarian and near-centenarian subjects and two control cohorts were determined. Results show in the centenarian cohort an increased frequency of subjects carrying the homozygous genotype for the functional variant of TAS2R38 (PAV/PAV) and a decreased frequency of those having homozygous genotype for the non-functional form (AVI/AVI), as compared to those determined in the two control cohorts. In conclusion, our data providing evidence of an association between genetic variants of TAS2R38 gene and human longevity, suggest that TAS2R38 bitter receptor can be involved in the molecular physiological mechanisms implied in the biological process of aging.
Introduction
The sense of taste is defined as a sensory system in which taste receptor cells are capable of detecting chemical molecules and provide valuable information about the nature and quality of food, but also about health-related issues1,2. In humans, taste receptors were originally identified and named based on their role in the taste cells of the tongue where they can discriminate five basic qualities: sweet, sour, salt, umami and bitter3. However, recent studies have shown that taste receptors are also expressed in a numerous extra-oral tissues throughout the body, including the airway and gastrointestinal tract, pancreas, liver, kidneys, testes, bladder and brain where they participate in a variety of physiological processes4,5,6,7,8,9,10. Although the role of taste receptors in the extra-oral tissues has been partially elucidated, recent studies have shown that they sense chemical molecules by means of transduction mechanisms and chemosensory signalling pathways like those occurring in the taste cells of the tongue5,8. Disorders or modifications in the sensitivity or expression of these extra-oral receptors and signalling pathways can affect physiological functions5.
Specifically, bitter taste receptors (T2Rs) that belong to the G-protein coupled receptors superfamily, detect many bitter chemicals with different chemical structures and plant-based compounds11. Humans possess approximately 25 bitter receptors (TAS2R)12. Traditionally, it has been assumed that they initiate bitter taste perception in the oral cavity which serves as a central warning signal against the ingestion of potential toxins. However, growing evidence indicates that T2Rs are widely expressed throughout the body where they mediate diverse non-tasting functions and that their genetic variants are associated with different human disorders9.
Among T2Rs, TAS2R38 has been widely studied because it mediates the bitter taste of thiourea compounds, such as phenylthiocarbamide (PTC) and 6-n-propylthiouracil (PROP), which has been reported as an oral marker for individual differences in taste perception, general food preferences and dietary behaviour, with consequent links to body mass composition and other non-tasting physiological mechanisms1,2,9,13,14,15,16,17. Several results show that perception of to the bitter taste PTC/PROP is associated with perception of other taste stimuli13,18,19,20,21,22,23,24,25, food preferences and choices13,26,27,28,29. Peculiarly, PROP super-taster individuals, compared to PROP non-tasters, seem to show a greater sensitivity and a lower liking and intake for high-fat/high-energy foods1,20,30, and a reduced intake of vegetables and fruits13,16,17,26,28,31,32. However, other studies did not confirm these associations33,34,35,36,37,38,39,40 suggesting that other factors may contribute to dietary predisposition and eating behaviour2. PROP perception has also been associated to health markers including: body mass index28,41, metabolic changes which impact on body mass composition42,43, antioxidant status44, colonic neoplasm risk45,46, smoking behaviors47, consumption of alcoholic beverages20, predisposition to respiratory infections48 and even neurodegenerative diseases49,50,51,52,53. In addition, TAS2R38 has been shown to detect bacterial quorum-sensing molecules and to regulate nitric oxide-dependent innate immune responses of the human respiratory tract48.
The gene codifying for TAS2R38 receptor is characterized by three non-synonymous coding single nucleotide polymorphisms (SNPs) which result in two common variants of the TAS2R38 protein: the functional form containing proline, alanine and valine (henceforth the associated genotype is named as PAV) and the non-functional variant containing alanine, valine and isoleucine, (with the genotype named as AVI)20,54. TAS2R38 SNPs dictate individual differences in PTC/PROP tasting54,55,56, food linking patterns1,57 and also in TAS2R38‒mediated pathophysiology9, such as susceptibility, severity, and prognosis of upper respiratory infection, chronic rhinosinusitis and biofilm formation in chronic rhinosinusitis patients48,58,59,60,61,62,63,64, development of colorectal cancer45,65, taste disorders66 and neurodegenerative diseases49.
Given the implications of TAS2R38 bitter receptor in taste perception, food preferences, diet and nutrition2 (which can affect human development and subsequently longevity67,68,69,70), and those in an efficient immune response48 and disease aetiology9,45,49,66 (which modulate the physiological mechanisms involved in the biological process of aging71,72), it is likely that TAS2R38 and its variants can play an important role in the attainment of longevity.
In this study we analysed the genotype distribution and allele frequency of the TAS2R38 gene in a cohort of centenarian subjects recruited in a genetically isolated area of the central-eastern Sardinia island (called the Longevity Blue Zone, LBZ). This area, which includes six mountainous villages of Ogliastra and Barbagia, has a total population of nearly 12,000 inhabitants on a land area of 888 km2 (Fig. 1)73. This population has remained isolated for many centuries, which made its genetic make‒up one of the most homogeneous in Europe74, and its sociocultural and anthropological characteristics well preserved throughout history73,75. LBZ represents an interesting case study because it shows a value of the Extreme Longevity Index (ELI)76 computed for generations born between 1880 and 1900 that is more than twice as high as that of the whole Sardinia. For this reason, data from this population were compared with data from ancestrally‒diverse cohorts recruited in another area of Sardinia (area of Cagliari, in the south of Sardinia).
Figure 1. Geographical location of the Longevity Blue Zone villages.
The association between longevity and taste genetics has been already investigated77,78. Campa and co-authors77 analysed, in a population from Calabria (Italy), the association with longevity of the common genetic variants of three bitter taste receptor genes that are involved in food preferences, food absorption processing and metabolism. They compared the results from centenarian subjects with those from non-elderly controls with a wide age range (20–84 years). Results showed that the frequency of subjects who carried the genotype homozygote AA for the polymorphism, rs978739, in TAS1R16 gene increases gradually from 35% in subjects aged 20 up to 55% in centenarians. However, another study did not confirm this association, by comparing results from centenarian with those from young controls (age range 18–45 years), in a population from another area of South Italy (Cilento) which could be subjected to different demographic pressures78. Here, we decided to compare results from centenarian subjects of the LBZ with those of two control cohorts south of Sardinia differentiated based on their age, one of young adult subjects (age ranging from 18 to 35 years) and a second of middle-aged adults and older adults (age ranging from 36 to 85 years).
.../...
Results
Molecular analysis of the TAS2R38 polymorphisms identified in the LBZ cohort (n = 94) 32 subjects who were PAV homozygous, 38 heterozygous, 17 AVI homozygous and 7 carried rare haplotypes (3 had AAV/AVI genotype, 2 PAV/AAV, 1 AAV/AAV and 1 AAI/AVI), in the CY cohort (n = 181) 41 subjects were PAV homozygous, 85 heterozygous, 48 AVI homozygous and 7 carried rare haplotypes (1 had AAV/AVI genotype, 2 PAV/AAV, 2 PVI/AVI, 1 PAV/AAI and 1 AAI/AVI) and in the CMAE cohort (n = 98) 18 subjects were PAV homozygous, 43 heterozygous, 35 AVI homozygous and 2 carried rare haplotypes (1 had PAV/AAV genotype and 1 AAI/AVI) (Supplementary information).
The three cohorts differed statistically based on their genotype distribution (Χ2 = 8.855; P = 0.0119; Fisher’s test) and haplotype frequencies (Χ2 = 11.31; P = 0.0035; Fisher’s test) (Fig. 2). Pairwise comparisons showed that the LBZ cohort differed from the other two cohorts (genotype: Χ2 > 6.381; P ≤ 0.041; Fisher’s test and haplotype: Χ2 > 6.96; P ≤ 0.030; Fisher’s test), which did not differ from each other (genotype: Χ2 = 2.667; P = 0.26; haplotype: Χ2 = 2.862; P = 0.239; Fisher’s test). The LBZ cohort was characterized by a high frequency of the diplotype PAV/PAV (34.04%) and haplotype PAV (55.32%) and a low frequency of diplotype AVI/AVI (18.08%) and haplotype AVI (40.42%), whereas the CY and CMAE cohorts were characterized by a lower frequency of diplotype PAV/PAV (CY: 22.65%; CMAE: 18.36%) and haplotype PAV (CY: 46.96%; CMAE: 40.81%) and a higher frequency of diplotype AVI/AVI (CY: 26.52%; CMAE: 35.71%) and haplotype AVI (CY: 51.10%; CMAE: 58.16%).
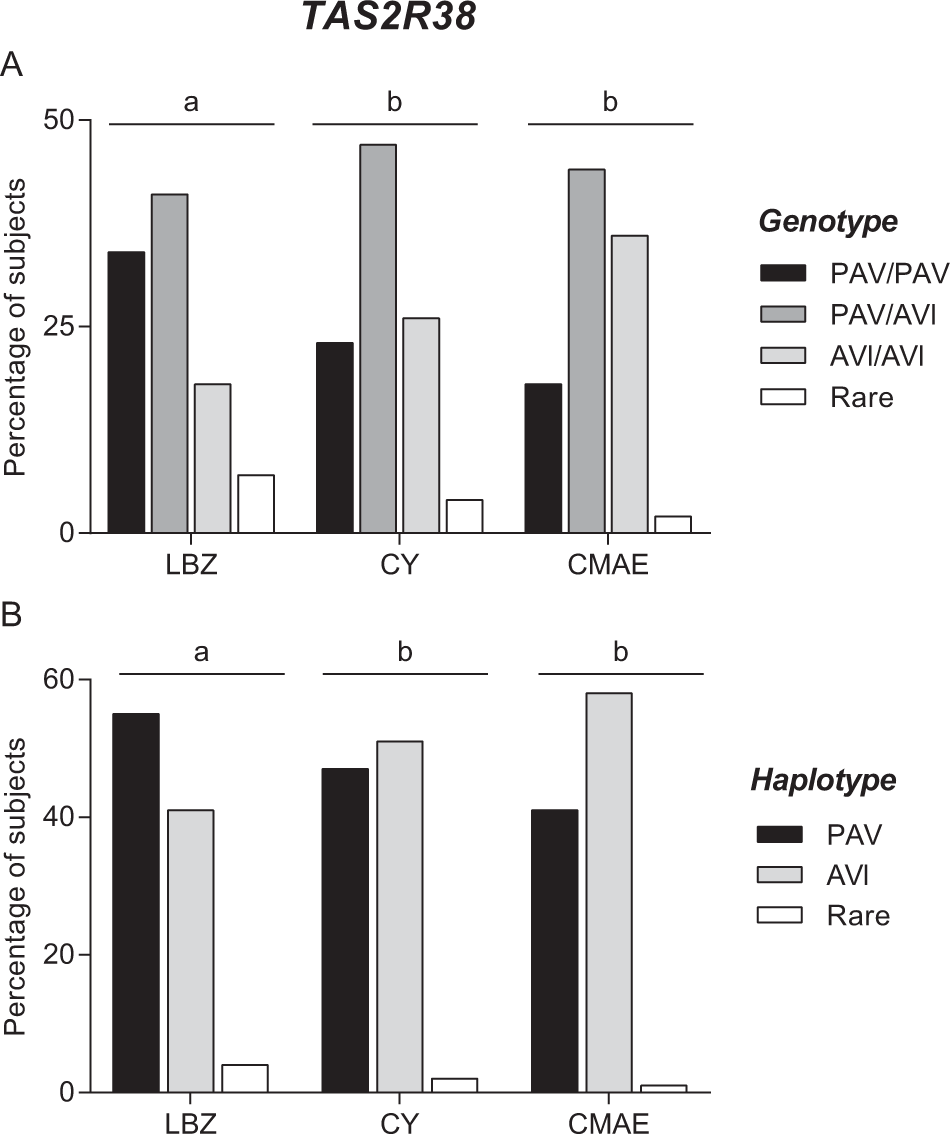
Figure 2. Genotype distribution (A) and haplotype frequencies (B) of polymorphisms of TAS2R38 gene in the Longevity Blue Zone cohort (LBZ) (n = 94), Cagliari young subjects’ cohort (CY) (n = 181) and the Cagliari cohort including middle-aged adults and elder adults (CMAE) (n = 98). Different letters indicated significant difference (Χ2 > 6.38; P ≤ 0.041; Fisher’s test).
.../...
.












































