.
O P E N A C C E S S S O U R C E : Aging us
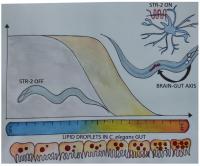
The landmark study by Kenyon et al. in 1993 showing doubling of the life span of Caenorhabditis elegans due to altered insulin-like growth factor signalling is considered by many as the first milestone in the longstanding quest for ‘longer life’ [1]. Decrease in growth factor signalling extends life span not only in worms but also in mammals. Since then, a number of genetic factors acting at cellular, tissue and organismal levels have been shown to regulate longevity and to ameliorate aging. This includes chromatin remodelling factors, anscription factors, enzymes involved in lipid metabolism and components of the cellular translation machinery and of autophagy. The actions of many of these factors converge on lipid metabolism in the intestine or the gut of C. elegans [2, 3]. However, it is not clear how various mechanisms for life span extension are coordinated in a multicellular organism? Is the brain of animals capable of coordinating and regulating longevity pathways? In 1999, an elegant study by Apfeld and Kenyon showed the role of sensory cilia in the control of life span in C. elegans [4]. However, the mechanism by which sensory neurons, numbering 48 in the adult nematode, exert control over aging remained elusive. Alcedo and Kenyon extended the study by systematically ablating Amphid sensory neurons [5]. Several of the ablated neurons were attributed with a role in regulation of longevity although each neuron had only a small impact. C. elegans sensory neurons express multiple G protein coupled receptors (GPCRs), and thus the ablation of an entire neuron may appear to have no net effect due to the absence of multiple GPCRs which may have opposing effects. Therefore, it is desirable to study individual GPCRs than entire neurons, to gain mechanistic understanding of longevity pathways. In a recent study we asked whether specific GPCRs, expressed only in the sensory neurons, exert control over tissue systems. By systematically examining the role of olfactory GPCRs in the regulation of longevity, we found that serpentine receptor STR-2 is involved in the regulation of longevity in C. elegans [6]. One of the surprising findings is that this olfactory GPCR, restricted to just three neurons, has a rather large impact on life span. Activity of STR-2 in olfactory neurons AWC and chemosensory neurons, ASIL and ASIR, was necessary for longevity on E. coli diet and at high temperature of growth above 20°C.
Edited by Engadin, 29 September 2020 - 05:58 PM.